Paper Battery
|
A paper battery is a flexible, ultra-thin energy storage and production device formed by combining carbon nano tubes with a conventional sheet of cellulose-based paper. A paper battery acts as both a high-energy battery and super capacitor , combining two components that are separate in traditional electronics . This combination allows the battery to provide both long-term, steady power production and bursts of energy. Non-toxic, flexible paper batteries have the potential to power the next generation of electronics, medical devices and hybrid vehicles, allowing for radical new designs and medical technologies.
Paper batteries may be folded, cut or otherwise shaped for different applications without any loss of integrity or efficiency . Cutting one in half halves its energy production. Stacking them multiplies power output. Early prototypes of the device are able to produce 2.5 volts of electricity from a sample the size of a postage stamp. What are carbon nano tubes (CNT)?
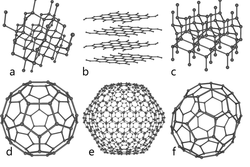
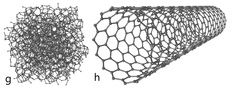
Carbon nano tube is an allotrope of carbon. Allotropy is nothing but different structural modifications of an element. For example the following image shows the different allotropes of carbon
As you can see from the figure all the allotropes have different arrangement of atoms. For example, in case of diamond, the hardest element found on the earth, every single carbon atom is linked or bonded (covalent bond) with every other 4 atoms of carbon. Likewise, carbon nano tubes are a one of the allotropes of carbon where every carbon atom is linked with every other three carbon atoms and also form a cylindrical structure. Carbon nano tubes are man made. The graphite and diamond are alone the naturally occurring allotropes of carbon.
|
Emerging Technologies |
Working of Paper Battery
A very brief explanation has been provided.
Cathode: Carbon Nano tube(CNT)
Anode: Lithium metal (Li+)
Electrolyte: All electrolytes (incl.bio electrolytes like blood,sweat and urine)
Separator: Paper (Cellulose)
Anode and cathode are the metal electrodes in the battery. The electrodes are placed in the battery in such a way that, they are in contact with the electrolyte. When an external load connected to the battery circuit is closed, current begins to flow because of the electro chemical reactions occurring inside the battery. Oxidation and reduction are the two main electro chemical reactions happening in any batteries. Oxidation takes place at anode and reduction takes place at cathode. Anode loses electrons to the ions from the electrolyte to form a compound and hence is said to be positively charged. Cathode gains electron from the electrolyte to form a compound and hence is negatively charged.
Cathode: Carbon Nano tube(CNT)
Anode: Lithium metal (Li+)
Electrolyte: All electrolytes (incl.bio electrolytes like blood,sweat and urine)
Separator: Paper (Cellulose)
Anode and cathode are the metal electrodes in the battery. The electrodes are placed in the battery in such a way that, they are in contact with the electrolyte. When an external load connected to the battery circuit is closed, current begins to flow because of the electro chemical reactions occurring inside the battery. Oxidation and reduction are the two main electro chemical reactions happening in any batteries. Oxidation takes place at anode and reduction takes place at cathode. Anode loses electrons to the ions from the electrolyte to form a compound and hence is said to be positively charged. Cathode gains electron from the electrolyte to form a compound and hence is negatively charged.
Construction of paper batteries
- A common xerox paper of desired length is taken
- A specially formulated ink (with CNT) is spread over the paper
- On the other side a thin film of lithium is laminated.
- Aluminium rods are connected for carrying current.
Benefits
|
Applications
|